 
Other animals, and which does not achieve its primary intended purposes U intended to affect the structure or any function of the body of man or 
U intended for use in the diagnosis of disease or other conditions, or in theĬure, mitigation, treatment, or prevention of disease, in man or other Pharmacopoeia, or any supplement to them, U recognized in the official National Formulary, or the United States Implant, in vitro reagent, or other similar or related article, including U "an instrument, apparatus, implement, machine, contrivance, Need to use medicines and foods to maintain and improve Public get the accurate, science-based information they Nation’s food supply, cosmetics, and products that emitįDA is also responsible for advancing the public healthīy helping to speed innovations that make medicines moreĮffective, safer, and more affordable and by helping the Veterinary drugs, biological products, medical devices, our U Additional tips & useful FDA databasesįDA is responsible for protecting the public healthīy assuring the safety, efficacy and security of human and U FDA’s authority & device classification What does the 510(k) pathway really look like?įDA 510(k) Submission Tips & Best Practices What happens to your 510(k) once it’s submitted to FDA? How to avoid having your 510(k) rejected What does the 510(k) timeline look like and how long is it going to take? What exactly are the expectations for your 510(k)? Insider tips, tricks and best practices to getting your device to market faster 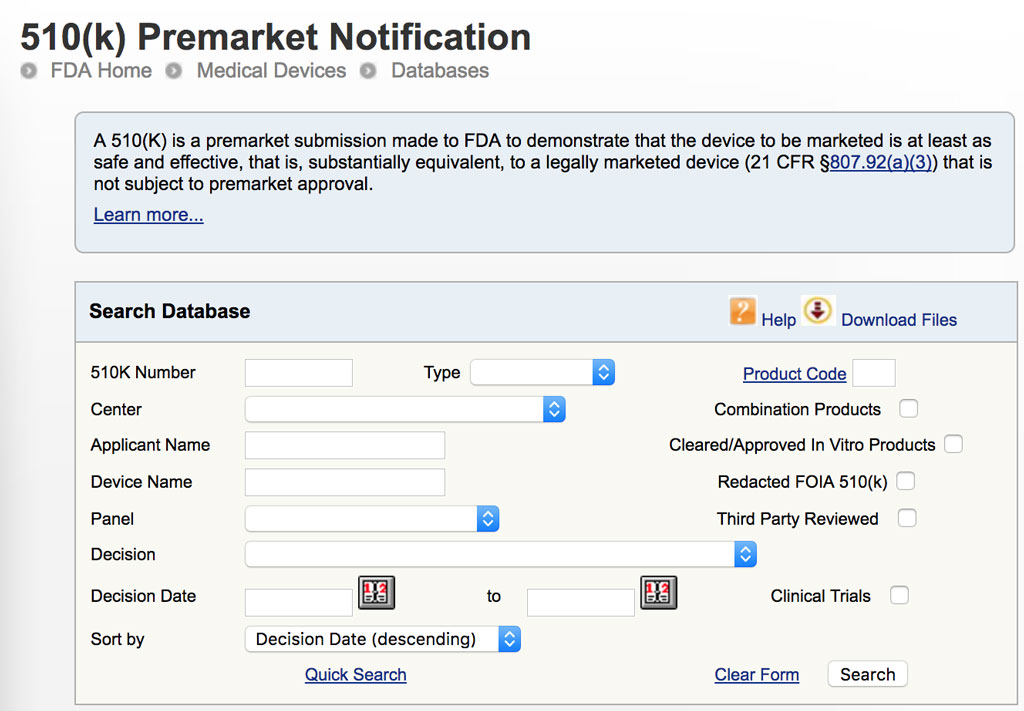
FDA 510K DATABASE FULL(You can view the full free webinar here: ) Join us for this free, 60 minute webinar, presented by our guest Allison Komiyama, PhD, RAC, principal consultant at AcKnowledge Regulatory Strategies and a former FDA reviewer.Īllison will be sharing with you some of her insider tips and best practices so you have a better chance at receiving your clearance letter faster than your competitors! How can you piece together your submission in a way that’s least likely to be rejected? So what can you do to better manage your team’s 510(k) expectations? These will be addressed on the next business day.Did you know that between January and June of 2015, 69% of 510(k) applications were rejected their first time?Įven though there have been significant changes to the content required in a 510(k) submission since the agency released their guidance in 2005, this should not be the case. You may leave a message or send e-mail at other times. Eastern Time (see Federal Holidays and Federal Government Operating Status). Government business days (Monday to Friday, excluding U.S. FDA 510K DATABASE REGISTRATIONTo e-mail questions about the Bioterrorism Act use this form Account Management Help Electronic Submissions Gateway HelpĮffective January 14, 2004: The Food Facility Registration Data Management Support Services (FFRDMSS) is available for technical assistance with online registration and listing systems, and regulated electronic submissions on U.S. FDA Industry Systems / FDA Unified Registration and Listing Systems (FURLS) / Technical HelpĮlectronic Submissions Gateway Approved Production Transaction Partners, Food Facility Registration Module, Low Acid & Acidified Canned Foods, and Account Management. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
